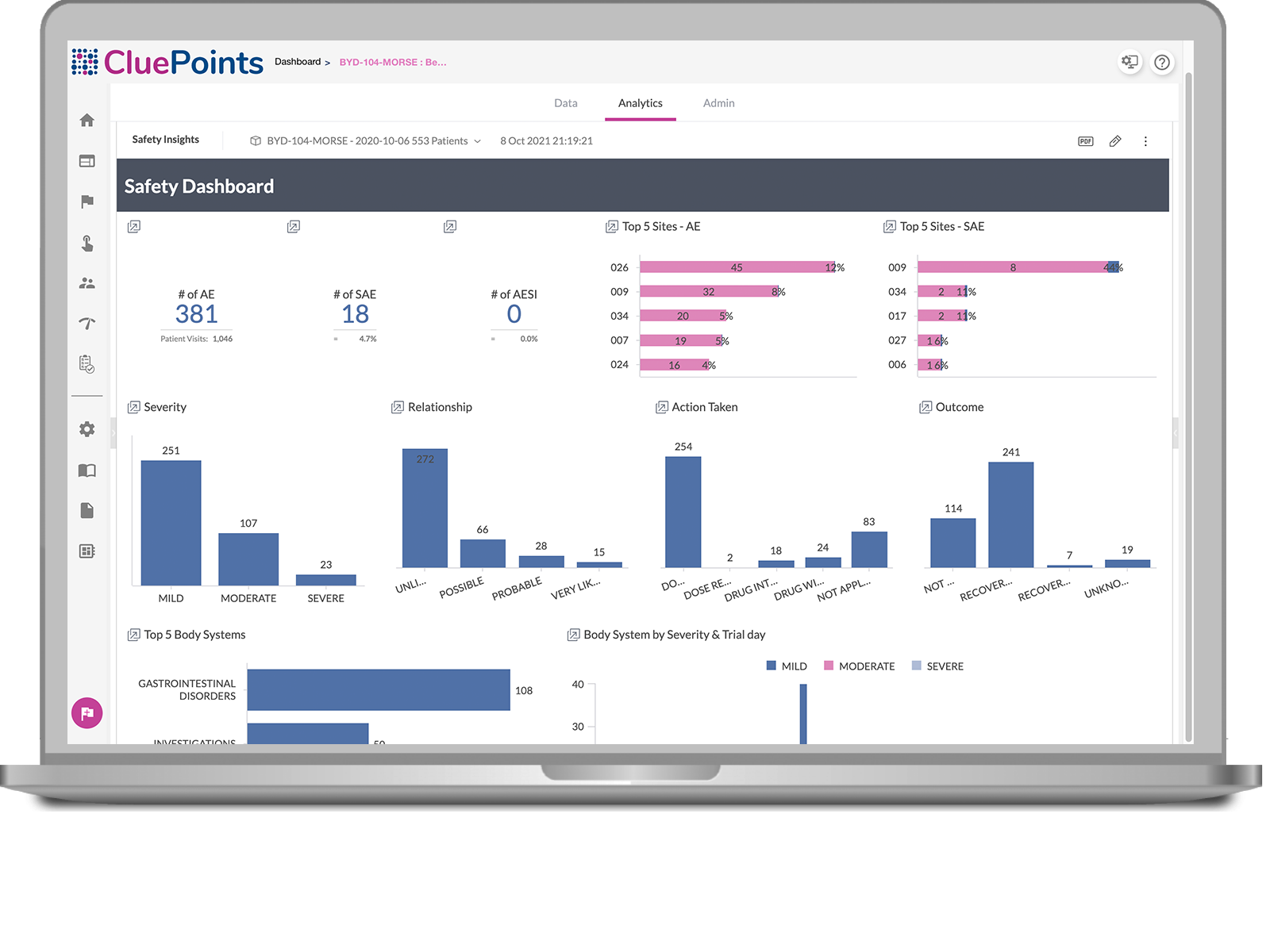
Adverse Event of Special Interest/AESI/AESI in Pharmacovigilance/AESI in CT/Pharmacy job interview - YouTube
Guide for Surveillance of Adverse Events of Special Interest (AESI) during Novel Oral Polio Vaccine Type 2 (nOPV2) Use

Challenges in conducting post-authorisation safety studies (PASS): A vaccine manufacturer's view - ScienceDirect
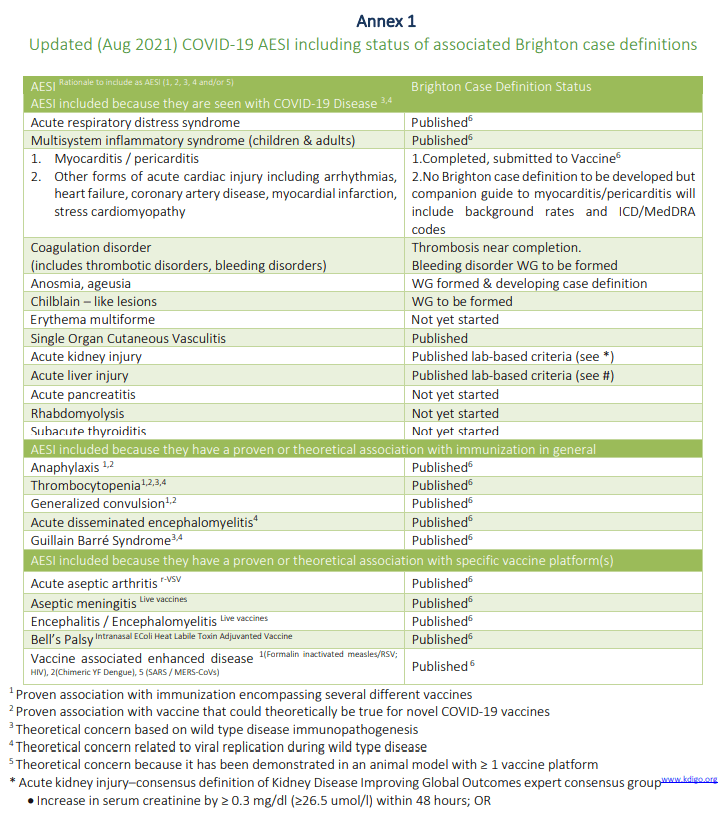
Peer review fail: Vaccine publishes antivax propaganda disguised as “reanalyses” of Pfizer and Moderna COVID-19 vaccine clinical trial data | Science-Based Medicine
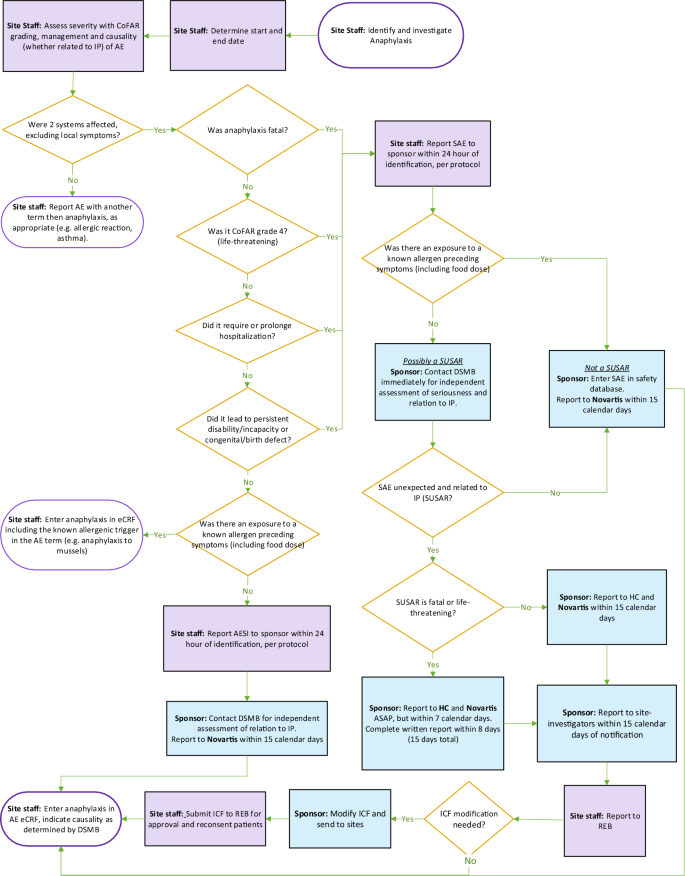
Protocol for a double-blind, randomized controlled trial on the dose-related efficacy of omalizumab in multi-food oral immunotherapy | Allergy, Asthma & Clinical Immunology | Full Text

Definitions and difference in PVG terms: Designated medical event (DME), Important medical events (IMEs), WHO critical terms and AESIs
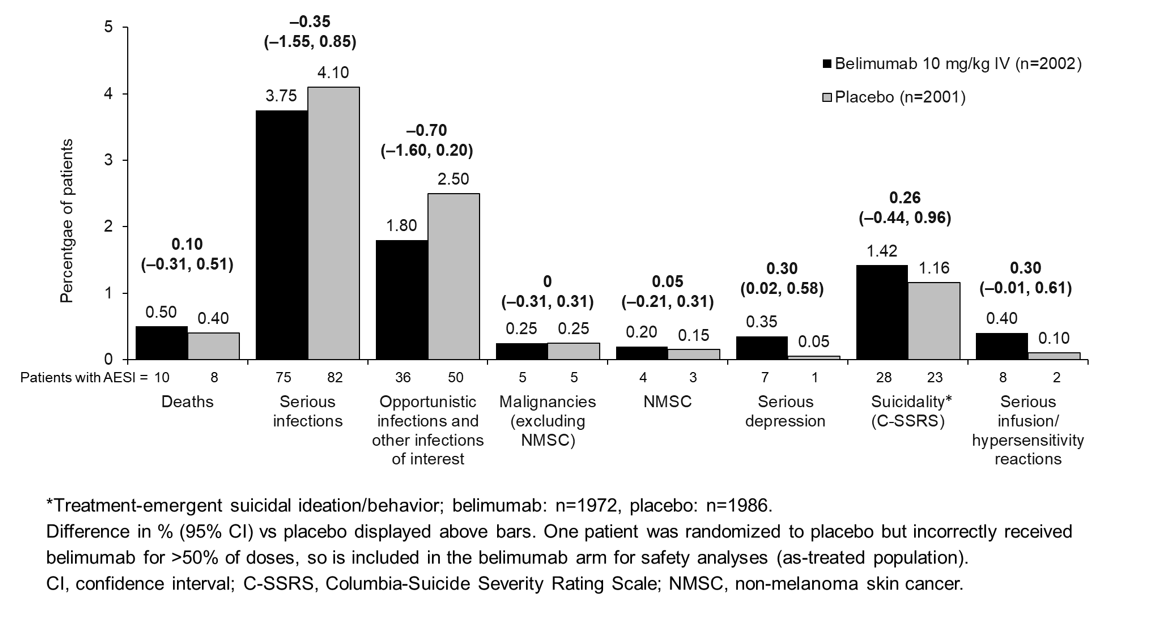
Adverse Events of Special Interest, SLE Medication Utilization, Hospitalizations, and Organ Damage: Results from a Phase 4, Randomized, Double-Blind, Placebo-Controlled, 52-week Study of Belimumab in Adults with Active, Autoantibody-Positive SLE - ACR